The probiotic that beats irritable bowel syndrome
Symprove® is scientifically proven to outperform other leading probiotic brands
Over the past few years, probiotics have become increasingly popular among health-conscious consumers for their wide range of preventive and therapeutic benefits, and are now a major sector of the nutritional supplements industry.
Considering that probiotics, or beneficial bacteria, support the immune and gastrointestinal (GI) systems, inhibit harmful pathogens, reduce the occurrence of allergies, and protect against a variety of chronic diseases, their popularity is not surprising. But with this rise of interest comes an ever increasing number of products on the market which range in quality from those that have been scientifically validated and tested in clinical studies, to those with unsubstantiated claims of potency and efficacy.
How do you know what you’re getting when you purchase?
In choosing a product, make sure it meets these criteria or quality standards:
-
- It must be demonstrated to contain sufficiently large numbers of live, viable bacteria which are resilient enough to survive transit through the hostile environment of the GI tract and form healthy colonies;
- It must be proven effective in providing a health benefit as demonstrated in a controlled clinical study;
- It must be manufactured according to strict quality control and purity standards, with each batch tested for identity and potency.
Of the hundreds of products on the market to choose from, one stands above the competition and meets each of these criteria: Symprove®, a high-quality, high-potency liquid product containing live active cultures of four probiotic strains. Let’s examine the wide-ranging benefits of probiotic supplementation and the importance of choosing a scientifically verified and clinically proven product.
Probiotics Defined
The term “probiotics” literally means “for life.” The United Nations Food and Agricultural Organization and the World Health Organization define probiotics as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.” (1,2) Note that the organisms must be delivered live and in sufficient numbers to the gut, and that they must produce some beneficial effect(s) on the host animal or human. As we’ll see below, Symprove® was confirmed to meet both parts of the technical definition of probiotics; not all products that are labeled “probiotic” meet these standards.
Probiotic bacteria are classified according to their particular genus, species, and strain. Within each genus are multiple species (for example, the genus Lactobacillus currently consists of over 125 species, such as L. acidophilus and L. rhamnosus), and within each species, different strains. Each species and strain has specific actions and uses.
Figure 1: Probiotic bacteria
Health Benefits of Probiotics
Probiotic cultures have been used for thousands of years, originally as a means of food preservation, in cultured milk products and fermented foods. Their health-promoting qualities have been widely touted for centuries, as in the case of the villagers of the Caucasus Mountains whose frequent consumption of yogurt has long been associated with good health and longevity.
Since the first clinical trials in the 1930’s, researchers have demonstrated the broad array of health benefits provided by probiotic supplementation. Beneficial bacteria may best be known for their role in contributing to GI health. They improve intestinal microbial balance, and enable the digestive system to function optimally. In fact, the intestinal tract is a mini-ecosystem (part of the human microbiome) ─ home to over a hundred trillion bacteria, from four to five hundred different species, in addition to a host of other microorganisms such as yeasts, fungi and protozoa. The key to good health is a positive balance of all of these “residents,” with sufficient numbers of beneficial bacteria performing all of their vital roles while keeping the less desirable pathogenic microbes in check.
Probiotics have been shown to be especially useful in the treatment of diarrhea resulting from antibiotic use (which can decimate intestinal bacteria, both the good and the bad), rotaviruses (in young children) and traveling. Certain probiotics, including some Lactobacilli, may relieve constipation as well as alleviate symptoms of lactose intolerance and malabsorption by producing lactase in the intestine and stomach. Probiotic bacteria have also had success in treating irritable bowel syndrome (IBS) and inflammatory bowel diseases such as colitis and Crohn’s disease. We’ll examine in detail below a clinical study in which Symprove significantly improved IBS-related symptoms.
Since the GI tract is intimately connected with the immune system, probiotics also play a role in the immune response. They inhibit the growth of a variety of dangerous microbes (including common bacterial pathogens such as Salmonella and Shigella) and enhance immune factors. Without a healthy microbial balance in the gut, the immune system can’t function properly to ward off infection.
Probiotics have been shown effective in such diverse conditions as colon cancer, where they may reduce the incidence and number of tumors, autism, urinary tract and respiratory infections, bacterial vaginosis, allergies, high cholesterol, high blood pressure, and diabetes.
And, among other numerous roles of probiotics is the synthesis of vital nutrients such as B vitamins and Vitamin K.
Sources of Probiotics: Food vs. Supplements
So, where can you obtain these beneficial bacteria? Good sources of probiotics include cultured milk products such as yogurt and kefir and fermented foods popular in Europe and Asia, such as pickled vegetables, sauerkraut, kimchi (a traditional Korean pickled dish) and miso. But as healthy as these foods may be, it’s impossible to know just how many live, viable organisms are in the portion you’re consuming. Clinical studies indicate that an effective dose of probiotics is in the range of one to 10 billion CFU’s per day. (A CFU, or colony forming unit, is a measure of viable, live bacteria which divide to form colonies.) Daily consumption is essential ─ most probiotics do not permanently inhabit the intestines. Commercial yogurt manufacturers generally list on the product’s label the species of probiotic bacteria used and the “Live Active Culture” seal, but not the actual number of cultures. Due to the inherent variability in manufacturing processes, it’s impossible to know exactly how many live bacteria reside in a container of yogurt (unless analyzed by a commercial lab), especially when you factor in storage and transit conditions, and shelf life. Yogurt and other cultured foods are a valuable addition to the diet, but in order to obtain the higher end of the recommended probiotic dose (10 billion CFU’s per day) to maximize health benefits, supplementation with a probiotic product is essential. But which one?
How to Choose a Product
As mentioned, there’s a plethora of probiotics on the market, in the form of liquids, tablets, powders, and capsules, and choosing one can be a daunting task, unless you can be sure it conforms to the three fundamental criteria outlined earlier. First, for a product to be effective, the bacteria it contains must be live and active, and in sufficient numbers when consumed. And the formulation must ensure that the bacteria will survive the various trips involved from the point of manufacture to your home, and then the more challenging voyage through your stomach to your colon, where they must multiply and form colonies. (We’ll refer to adequate potency and viability of the bacteria in a probiotic product as Criterion 1.) Second, the product should be evaluated for efficacy in a clinical study; i.e., it must provide a measurable health benefit(s) (Criterion 2). And third, the product must be manufactured with high quality control and purity standards (Criterion 3). You really can’t be sure what you’re getting unless the product conforms to these quality standards.
Most Probiotics Don’t Deliver, but One Does
To provide much needed independent and comparative scientific data on the potency and quality of commercially available probiotic supplements (in other words, to address Criterion 1 on our list), scientists from the University College London School of Pharmacy conducted a study on eight commercial brands. Using test conditions that simulate the human GI tract, the researchers conducted three “challenge tests” to investigate whether the bacteria in each product (1) were present in the amounts stated on the label; (2) survived transit through the stomach; and (3) had the potential to form healthy colonies after exposure to gastric fluid. The eight probiotic brands were Actimel®, Align®, Symprove®, Biobalance®, Probio7®, Yakult®, VSL#3®, and Bio-kult®.

Figure 2: Comparison with other probiotics
In the first test on label claim, only liquid products (dairy-based Actimel® and Yakult® and water-based Symprove®) contained 100 percent or more of the labeled content of live bacteria. Freeze-dried, or lyophilized, products scored lower recoveries of live organisms; in these formulations, bacteria must rehydrate to return to a metabolically active state, which often results in the loss of numbers of organisms. Liquid products, on the other hand, contain bacteria that are already in an active state and scored higher recoveries.
In the second challenge test, the eight probiotic brands were submitted to simulated and porcine gastric fluids to mimic the acidic conditions of the human stomach. Most products showed a sharp decline in the number of bacteria after only five minutes in an acidic environment. Only two products contained bacteria that survived a full two hours (equivalent to stomach transit time) in gastric fluid, one (Symprove®) more robustly than the other (VSL#3®).
The third challenge test evaluated whether the bacteria in each of the products could grow and multiply within 10 hours after exposure to gastric fluid, a measure of their potential to colonize in the human GI tract. Only three of the eight products showed signs of strong growth within 10 hours (Actimel®, Symprove®, VSL3®).
Again, in order to be effective, a probiotic product must pass all three challenge tests: it must arrive alive (i.e. in sufficient numbers when consumed), survive stomach acid, and then thrive, or colonize in the intestines. If a product fails just one of these tests, it will be worthless (and four products failed all three!). Symprove was the only product that passed all three challenge tests, the equivalent of Criterion 1 on our list.
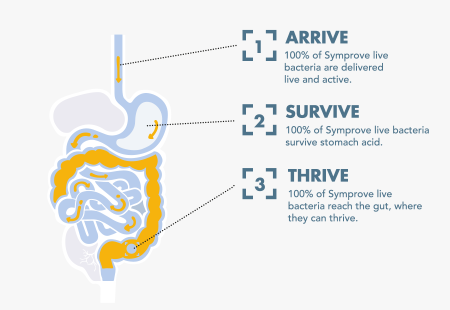
Figure 3: Symprove® passed all three challenge tests
Symprove’s unsurpassed performance in these in vitro experiments underlies its efficacy in vivo: in a controlled clinical study, it significantly alleviated troublesome symptoms associated with IBS, satisfying Criterion 2.
Symprove Effective in Treating IBS
IBS is a GI disorder characterized by abdominal pain, bloating, and bouts of diarrhea and/or constipation (not to be confused with inflammatory bowel disease). The condition is estimated to affect up to 15 percent of the population worldwide, and due to its problematic symptoms, results in reduced quality of life. Multiple factors may contribute to IBS, such as stress, depression, food sensitivities, hormonal changes, and imbalanced gut flora. Conventional treatments include drugs that are often ineffective in the long-term and laden with side effects. Because probiotics improve intestinal microbial balance, they are a promising alternative treatment, free of side effects.
In a 12-week placebo-controlled double-blind study at King’s College Hospital London, 186 patients with moderate to severe IBS received either Symprove® or a placebo, 1 ml/kg (which corresponds to approximately 68 ml for a 150-lb person or 55 ml for a 120-lb person) daily. The degree of symptoms, such as pain, constipation, diarrhea, and bloating, was measured at the beginning and end of the trial using a clinical tool known as the IBS symptoms severity score. By week 12, researchers noted a statistically significant improvement in overall symptom severity in patients who received Symprove® versus placebo. In most treated patients, pain, the predominant IBS symptom, as well as constipation and diarrhea, were reduced; by the end of the study, a quarter of these patients experienced minimal or no symptoms, an impressive outcome, especially as the patients were suffering from moderate to severe IBS. In addition, the product was well tolerated, even in these patients with pre-existing GI disturbances. (3)
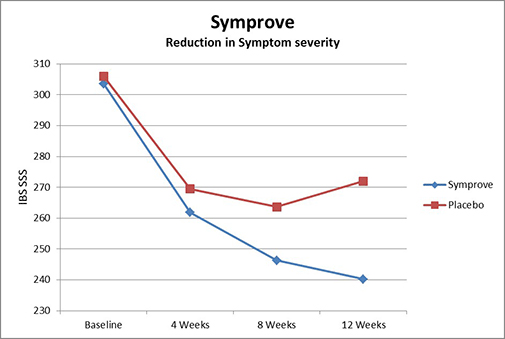
Figure 4: Improvement in IBS symptom severity with Symprove®
Bear in mind that probiotics are classified as foods, and so are not subject to the rigorous testing required for drugs. However, what’s noteworthy about this study, besides demonstrating Symprove’s efficacy in the human gut, is that it followed the standards equivalent of a drug trial, (i.e., the study design was randomized, double-blind, placebo-controlled).
Future research is underway to evaluate the efficacy of Symprove® in treating inflammatory bowel disease and diverticular disease.
What exactly is Symprove®
Symprove® is an aqueous extract of germinated barley containing live active cultures of four probiotic species: L. rhamnosus, E. faecium, L. plantarum, and L. acidophilus. Germinated barley extract acts as a substrate or prebiotic, helping the bacteria grow. Symprove’s formulation, or “Unique Delivery System,” protects and transports live probiotic bacteria safely through the stomach to the target area of the colon. Again, the bacteria in the product are live, which means actively metabolizing and dividing. The product is manufactured in a next-generation facility in the UK, in a clean-room environment to ensure high purity bacterial cultures. Each batch of Symprove® is analyzed by an independent lab to verify the identity of the bacterial formula and potency of 10 billion CFU’s per 50 ml serving (the upper recommended daily dose). These high standards for purity, identity, and potency satisfy Criterion 3.
How to Use Symprove®
Symprove® is available in a four-week pack containing four bottles. Each 50-ml serving can be measured in the cup provided and should be taken every morning on an empty stomach, 10 minutes before eating or drinking. Those experiencing GI symptoms are likely to see relief within four weeks, but it is recommended to use for a full 12 weeks to gain maximum benefits and rebalance gut bacteria. After 12 weeks, Symprove® may continue to be taken as part of a maintenance program.
Summary
We’ve seen that Symprove® meets the three required criteria for a superior probiotic supplement; it is a scientifically validated, clinically-proven, and quality-tested product that supports a balanced and robust population of gut flora to ensure the health of your GI system and your overall health.
Clearly anyone with IBS will, after seeing the results of the British clinical study, want to try a 12-week course of Symprove for themselves. But also, for those with general GI issues and even for those without- who wish to promote optimal health, or counteract a course of antibiotics, Symprove® is the probiotic of choice.
References
1. Sanders ME. Probiotics: definition, sources, selection, and uses. Clin Infect Dis. 2008 Feb 1;46 Suppl 2:S58-61; discussion S144-51.
2. Fredua-Agyeman M, Gaisford S. Benef Microbes. Comparative survival of commercial probiotic formulations: tests in biorelevant gastric fluids and real-time measurements using microcalorimetry. 2015 Mar;6(1):141-51.
3. Sisson G, Ayis S, Sherwood RA, Bjarnason I. Randomised clinical trial: A liquid multi-strain probiotic vs. placebo in the irritable bowel syndrome–a 12 week double-blind study. Aliment Pharmacol Ther. 2014 Jul;40(1):51-62.

